Becoming a Mother Shouldn’t Put Your Life at Risk: How market shaping can reduce global maternal deaths
This Mother’s Day, as we honor the women who raised us, we should also take a moment to reflect on a shocking fact: approximately 800 women die every day from complications related to pregnancy and childbirth.
Since 1990, maternal mortality has declined by 45 percent around the world. However, it is also noteworthy that, at the end of the Millennium Development Goals (MDG) era just 5 months ago, the goal of reducing maternal mortality by 75 percent had seen the least progress.
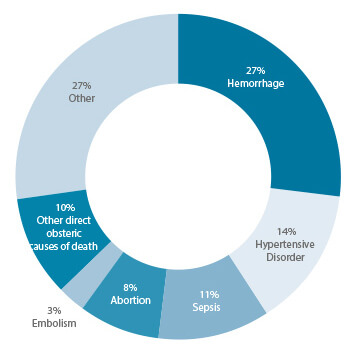
Globally, the two leading causes of maternal mortality are post-partum hemorrhage (PPH), excessive blood loss following the birth of a baby, and pre-eclampsia/eclampsia (PE/E), a complication marked by high blood pressure and protein in the urine; together, they comprise 40 percent of all maternal deaths. Three drugs can help prevent these deaths: oxytocin, misoprostol and magnesium sulfate. Oxytocin and misoprostol are both used to prevent and treat PPH, while magnesium sulfate is the most effective agent for treating seizures associated with PE/E. However, complex market barriers limit access to these life-saving drugs in low- and middle-income countries.
In order to reduce maternal deaths, the Reproductive Health Supplies Coalition (RHSC) commissioned Results for Development Institute’s (R4D) market dynamics team to scope market-based activities and identify key gaps and opportunities where market shaping interventions could help strengthen the availability of high quality, affordable oxytocin, misoprostol and magnesium sulfate. This market analysis was conducted at the global level and in three focus geographies—Ethiopia, Nigeria, and Bangladesh.
Our findings and recommendations were published in the report, Increasing Access to Essential Maternal Health Supplies.
The term “market shaping” refers to a proactive and holistic approach to catalyze activities explicitly intended to address market failures that are preventing desired key health outcomes. This includes working with all levels and actors in a marketplace, including manufacturers, donors, governments and buyers to improve the affordability, availability, quality, and market introduction timelines of appropriately designed medicines.
Based on the insights gathered from experts and in-depth analysis of data collected, we made several recommendations to address supply- and demand-side inefficiencies. Due to the complex barriers in these markets, the suggested interventions are multi-pronged and require national and sub-national engagement.
Supply-Side Recommendations
- Increase the number of high-quality verified products registered at the country-level.
- Conduct research to identify and scale up cost-effective tools to combat substandard and counterfeit drugs.
- Provide guidance on usage and improve product appropriateness by ensuring the design of maternal health supplies are appropriate for low-resource settings.
Demand-Side Recommendations
- Strengthen country-level data collection systems, such as the National Demographic and Health Survey and Civil Registration and Vital Statistics.
- Improve forecast development and linkages to supply plans.
- Reduce lead times and coordinate procurements, when possible.
- Increase market transparency on price ranges.
While there are no short-term fixes, it is important for partners in the maternal health space and across health sectors to coordinate efforts in tackling these challenges. Together, we have the opportunity to meet the 2030 development targets and achieve the Sustainable Development Goal of reducing the global maternal mortality ratio to less than 70 deaths per 100,000 live births.
We hope this report will contribute to more efficient markets for essential maternal health supplies and, in turn, a dramatic reduction in mothers’ deaths.
Read the full report, which was made possible with generous funding from the John D. and Catherine T. MacArthur Foundation.